Keywords:
By Anne Hartenhauer
“Myasthenia gravis is eminently treatable,” say researchers at UCL’s Institute of Neurology1. Yet clinicians still find it challenging to detect and manage. In a new webinar entitled “Autoantibodies in Myasthenia Gravis,” Dr. Jan Damoiseaux, a Laboratory Specialist in Medical Immunology at Maastricht UMC+ in the Netherlands, explores the underlying mechanisms of the disease and explains how a two-assay strategy can improve the accuracy of determination of autoantibodies and help monitor therapeutic efficacy to improve patient outcomes.
What is considered “best practice” for myasthenia gravis diagnosis and treatment monitoring? In our new webinar, Dr. Jan Damoiseaux explores the underlying mechanisms of the disease, and the benefits of a two-assay strategy.
Myasthenia gravis detection and quantification: rising to the challenges
Myasthenia gravis is an autoimmune disease that causes muscle weakness and fatigue and can be challenging to diagnose. Gold standard biomarkers are available to quantify the autoantibodies expressed by most patients with myasthenia gravis. In our new webinar, Dr. Damoiseaux illustrates what makes myasthenia gravis an autoimmune disease. He describes the two main forms of the disease, and the mechanisms by which the disease-related autoantibodies cause symptoms.
Most importantly, Dr. Damoiseaux discusses the best practices for myasthenia gravis diagnosis based on detection and quantification of the two types of disease-related autoantibodies. He provides a detailed explanation of the dual assay strategy used to diagnose the two forms of myasthenia gravis, and presents patient examples to illustrate the importance of quantitative assay results that can be used to guide patient monitoring and therapeutic decision-making. In addition, Dr. Damoiseaux briefly reviews the therapeutic options for patients with myasthenia gravis.
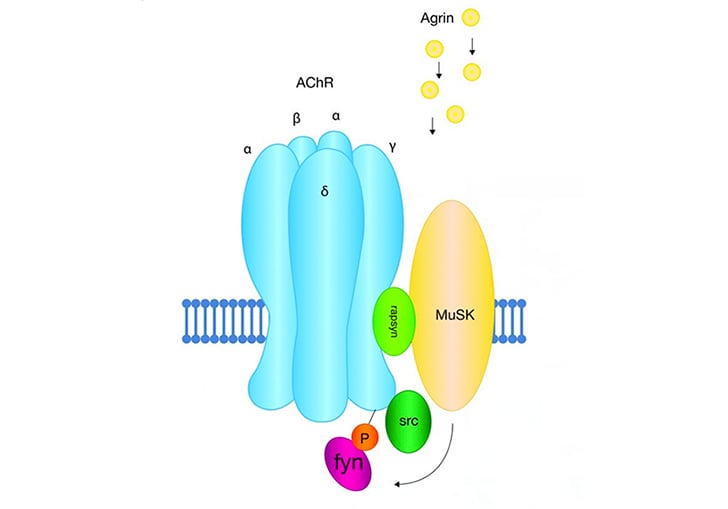
The two-assay strategy: ARAb and MuSK autoantibody determination
The initial step in diagnosing myasthenia gravis requires a radioreceptor assay (RRA) to detect acetylcholine receptor autoantibodies (ARAbs). An ELISA is not specific enough and will yield too many false positive results. Dr. Damoiseaux’s team uses the ARAb RRA from Tecan (IBL International), which incorporates α-Bungarotoxin from snake toxin to increase specificity.
Why is it so important to obtain quantitative ARAb RRA results? The level of antibody may vary widely between patients. While this does not relate to patients having a more or less severe form of the disease, it does have important implications for achieving remission. The target for remission in patients with ARAb-positive myasthenia gravis is about a 50% reduction in ARAbs, regardless of the starting value.
For patients with seronegative myasthenia gravis, the next step is to test for muscle-specific tyrosine kinase (MuSK) autoantibodies. Patients with MuSK autoantibodies tend to have a more severe form of the disease, and about 30% require respiratory support. Dr. Damoiseaux’s team uses a Tecan MuSK-Ab ELISA that yields quantitative results (IBL International). Identifying this group of patients – including screening ARAb-positive patients -- can lead to earlier initiation of effective treatment and improved quality of life. The MuSK-Ab ELISA can be used to monitor therapy, which ensures the complete elimination of MuSK autoantibodies that is required for remission.
To learn more about Dr. Damoiseaux’s work, watch the Tecan webinar “Autoantibodies in Myasthenia gravis.”
References
1 Spillane J, Higham E and Kullmann DM. Easily missed? Myasthenia gravis. British Medical Journal (2013) 346:36-39. https://www.bmj.com/bmj/section-pdf/187724?path=/bmj/346/7891/Practice.full.pdf
Tecan product expert
 |
Dr. Constanze Drechsel joined Tecan in 2019 as a product manager responsible for the autoimmunity and infectious disease portfolio. Constanze has a scientific background and completed her PhD research in protein and allergy chemistry and now supports the Global Reagent Marketing & Support department with immunoassay solutions at Tecan.
|
Dr. Constanze Drechsel |
Keywords:
About the author

Anne Hartenhauer
Anne Hartenhauer joined IBL International / Tecan in 2016 as product manager. She was responsible for Immunoassays in the clinical diagnostic fields infectious disease and autoimmunity.











