By Nick Smith
The launch of your new in vitro diagnostics (IVD) system is imminent. You’ve read in our first article about the 5 major ingredients for success: retaining technical expertise, investing in training, ensuring regional focus, forecasting accurately, and managing the lifecycle of your platform. Let us now re-examine these 5 ingredients to see how you can best use the support of your OEM partner to simplify the launch process, accelerate revenue ramp-up, and position your platform for long-term profitable growth.
-1.jpeg?width=768&height=512&name=AdobeStock_594268670%20(1)-1.jpeg)
OEM partnering for automated lab system development: 5 steps to speed up time to revenue
1. Leverage your OEM partner’s technical expertise
Your core development team experts will include key contacts with your OEM development partner as well as your early adopter customers. Keeping this team together for at least six months after launch will result in the growth of a valuable knowledge resource for your broader team, from customer service to service engineers.
Ideally you will have worked through the development process with an OEM partner that has a solid legacy of automation expertise as well as an in-depth understanding of your applications and their potential markets. If your company aspires to be a successful athlete in the diagnostics race, then that automation expertise must include a modular approach to enable accelerated application development and system testing. And whilst your team will of course have a thorough knowledge of the application/s being developed, the fact that your OEM partner also has application expertise means that together you will be able to create the tailored solutions you need in the shortest possible time.
In conclusion, whether you are working on automating workflows in immunodiagnostics, molecular diagnostics, tissue diagnostics, mass spectrometry sample preparation or any number of other applications, make sure that you choose an OEM partner that (1) uses agile, modular development methods, and (2) understands your applications and their objectives (see Fig. 1).
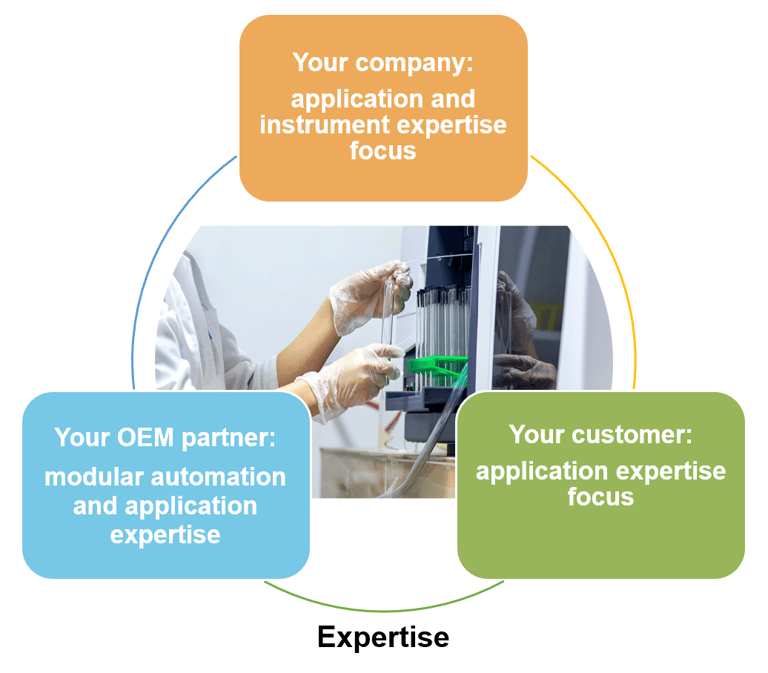
Figure 1: work with your OEM partner to leverage their automation and application expertise
2. Invest in training your employees and customers
Prior to and during launch, you will be giving standardized and assessed product and application training to all functions, tailored to the right level, preferably with some form of recognition of people’s training achievements. Making a similar programme of formal training available to your customers is also an excellent way to reduce the risks associated with user error, and should be an essential part of the installation process. If you have larger teams, of say 10 or more people, you can also try a “train the trainer” approach, with allocated “champions” within those teams.
In principle, the more people and functions that receive basic product information and training, the more successful will be the post-launch ramp-up. Engagement in your organization can be increased by making sure that all functions have a very basic knowledge of what your new IVD system is supposed to do, and a simple explanation of what impact it will have in the diagnostic world, with higher levels of training being reserved for specific functions.
For example, with their instrument and applications knowledge, the key contacts in your OEM partner team are ideally positioned to give higher level training to your service engineers and application specialist teams. Since your IVD instrument will have been developed on a modular basis, bringing knowledge of those modules in-house without the hassle of having gone through the entire development process, your teams will have more time to focus on training customers and on helping them optimize their applications (see Fig. 2).
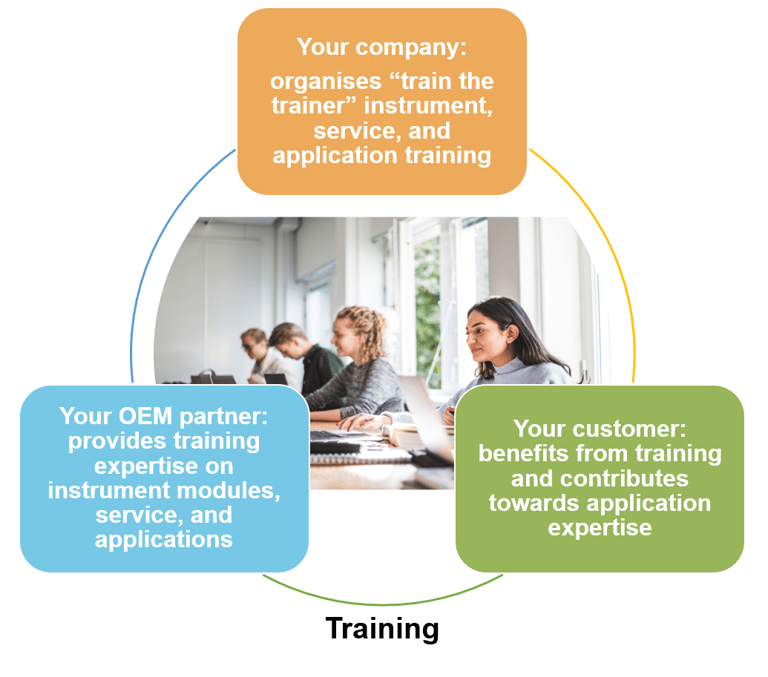
Figure 2: work with your OEM partner to provide a comprehensive training programme within your organization and for your customer
Taken to its logical conclusion, investing in learning and development in this tailored fashion will develop your employees’ understanding of your company's goals, and may even lead to higher productivity, as they acquire the skills, knowledge and the confidence necessary to execute their daily tasks. When employees receive proper training, they are more likely to provide better customer service and information, so customers are more likely to make informed choices in your favour, ultimately leading to increased revenue. This realization is a good argument for training to be budgeted adequately as part of your launch strategy and post-launch follow-up.
3. Work on forecasting with your OEM partner
Working with an OEM partner who understands the diagnostic market can help you calculate realistic forecasts according to market needs in particular application areas. Even if you feel you have a similar or higher level of knowledge of a particular market, or have conducted conclusive voice-of-customer research, it may still be useful to sanity check your forecasts with your OEM partner.
Accurate forecasting is particularly important when there is a consumables stream for your instrument, especially if you depend on an external supply chain and limited expiry dates. Even if you are making your consumables kits completely in-house, accurate forecasting is essential to guarantee a robust supply chain of raw materials. Effective project management must not be underestimated when it comes to developing, testing and manufacturing consumables kits within the requisite timeframes. We can probably all remember a case where an instrument might have been functioning perfectly well, but the availability of consumables turned out to be the limiting factor. It may be that your OEM partner can also help take out some of the risk associated with that process at the point where you are sure of the kit content and necessary specifications, if they themselves develop consumables (see Fig. 3).

Figure 3: minimize servicing and supply chain risks by working with your OEM partner towards robust forecasting
Your OEM partner will have helped you carry out due diligence regarding instrument robustness as part of the system development process. They will hence be able to give you clarity on any potential teething issues, and even a suggested list of spare parts for service engineers to have readily available during the launch phase. This approach can significantly improve repair times if there are teething problems with your instrument, and minimise downtime for customers, which is especially important when you are at that tricky ramp-up stage between nurturing reference labs and establishing an installed base.
4. Use your OEM partner’s regulatory knowledge for geographical focus
Regulatory and registration requirements differ vastly across the globe when it comes to IVD testing and instruments. Again it is useful to work with your OEM partner here, since they are likely to have a pretty good knowledge of the regulatory requirements in the different countries and regions where you intend to launch and implement your roll-out plan.
Having a phased roll-out plan, starting in a region where you are sure of having a combination of established reference customer expertise, readily available sales and technical support, OEM partner coverage, and a robust supply chain for both instrument and consumables, all of which will help maximize your chances of a successful ramp-up (Fig. 4).
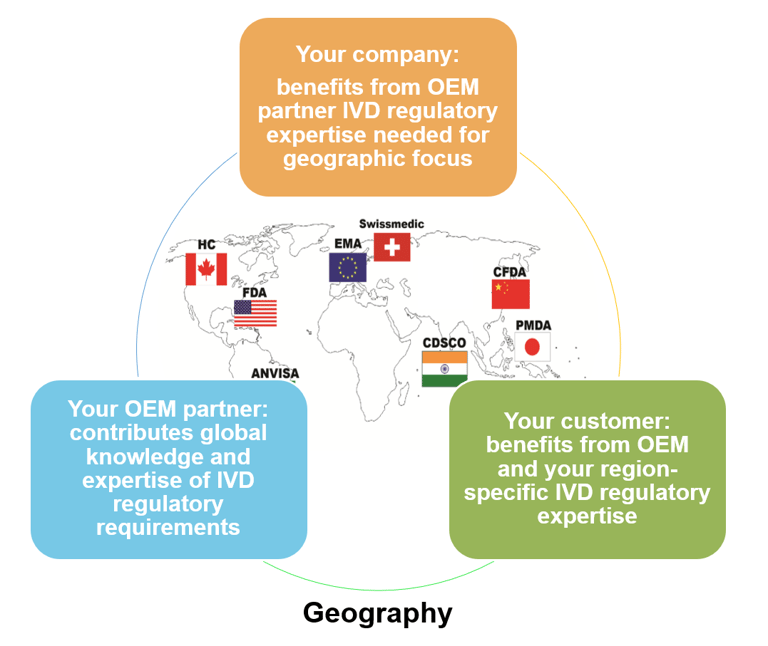
Figure 4: minimize geographically-specific regulatory risks by working with an OEM partner that has global and regional IVD regulatory expertise
5. Manage lifecycle effectively
In our first article we recommended that you implement a dedicated process for product lifecycle management, so that expected events can be planned for, and reaction time can be optimized for unexpected events. Product lifecycle can be simply expressed as several broad phases: development, introduction, growth, maintenance, and end of life. To help you plan how you will support your platform after launch, through ramp-up and beyond, we have developed a practical guide to product lifecycle management, which you can download here.
When it comes to ensuring ramp-up of your revenue during the growth phase, post immediate launch, you must make sure that your product meets with quality compliance specifications, that service and support levels are still delighting your customer, and that the platform is future-proof, with plans to implement product improvements and evolve according to market trends and needs, whether working towards a better price point or towards a new diagnostic assay.
A strong product management team is critical to effective product lifecycle management, collaborating across the organization and with your OEM partner, liaising with internal functions such as R&D, Quality, Operations, and indeed the customer, to help grow the instrument knowledge base and streamline the lifecycle management process (see Fig. 5).
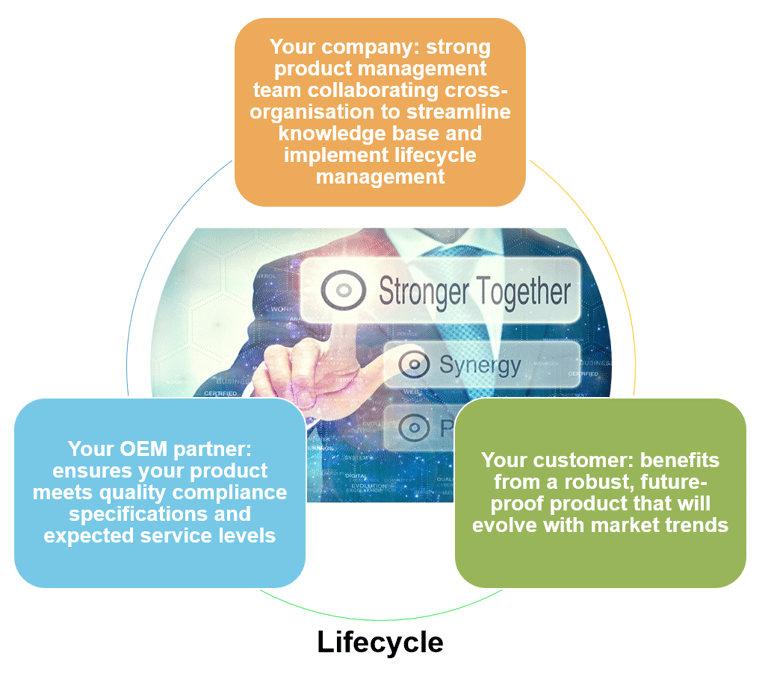
Figure 5: use OEM products that meet required quality compliance specifications so that product lifecycle can be effectively managed
That’s a lot to get right: how would you summarise it?
Every IVD system and launch plan is different, even if some of the workflows are similar, whether for immunodiagnostics, molecular diagnostics, tissue diagnostics, or mass spectrometry sample preparation, to name but a few. However, the guiding principles for a successful launch and revenue ramp-up are rather similar, so we have collected them into a white paper, together with some useful hints and tips, to help you reach one step further towards adapting and thriving in the ever-evolving diagnostics landscape.
About the author

Nick Smith
Nicholas Smith is VP Head Marketing for Tecan’s Partnering Business based in Switzerland. Nick has over 35 years of experience in the diagnostics industry and has held a variety of roles in multinational IVD companies both in Europe and in Asia. A bacteriologist by education, Nick holds a Fellowship of the Institute of Biomedical Science in the UK.











