Keywords:
By Nastya Yeska
Female hormone levels have been measured using blood tests for decades.1 However, blood sampling is costly, invasive, and often logistically difficult, so there is a shift towards the adoption of tests based on more convenient and cost-efficient sample types, such as saliva. Saliva-based tests are considered a reliable and proven method for measuring female hormone levels, as well as being highly accurate and painless for the patient.1 In this blog series, we look at how saliva testing for female hormone imbalance is changing the clinical diagnostics landscape, and positively impacting women’s health across the globe.
 Saliva testing is a sensitive, cost-effective method to diagnose a hormonal imbalance at any stage of a woman’s life.
Saliva testing is a sensitive, cost-effective method to diagnose a hormonal imbalance at any stage of a woman’s life.
Why do we need to measure female hormone levels?
Mention female hormone levels, and it evokes a whole spectrum of conditions that the medical profession has come to recognize as being real and treatable. Whether dealing with the more subjective mood and body changes associated with premenstrual syndrome, the heart-break of infertility, or simply remaining healthy through menopause, managing hormone imbalance is crucial, and can be a key contributor to a woman’s quality of life.2-4
Which female hormones do we need to measure?
The symptoms, syndromes and diseases experienced by women throughout their reproductive years are frequently due to hormone imbalances. These imbalances can be measured and treated by direct administration of the hormones that are lacking, or their precursor equivalents.5 Hormone treatments are often given, alongside recommendations on lifestyle changes, such as diet and exercise, to maintain or improve physical and cognitive health. You can read more about female steroid hormones in our guide.
Steroid Pathways
Fig. 1 shows a much simplified version of the interrelations between the different hormonal pathways in women: measurement of a selection of hormones within these pathways will give a medical professional an indication of any hormone imbalances, and lead to a recommended course of treatment.
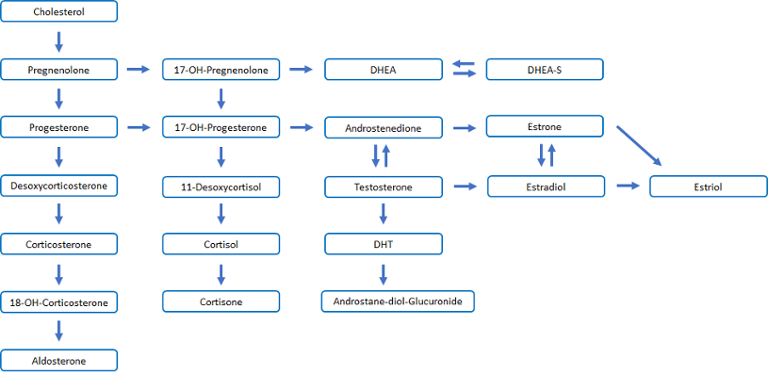
Alongside lifestyle changes, simple medicinal adjustments to hormone levels can make a huge difference to a woman who is suffering from a diagnosed hormone imbalance.
For example, during her reproductive years, a woman may suffer so greatly from symptoms related to premenstrual syndrome (PMS), or period-related problems, such as excessive bleeding, that her quality of life can be severely compromised.6 Most women suffer from PMS at some point in their lives, and about 30% of women globally find that the symptoms negatively affect their day-to-day activities.6
The menstrual cycle itself is influenced by various hormones: the female sex hormone estrogen is responsible for the maturation of one egg cell per month, and the lining of the womb prepares for a possible pregnancy, with the help of the hormone progesterone. Alongside other treatments, such as nonsteroidal anti-inflammatory drugs (NSAIDs) to help relieve pain, progesterone can be used to help reduce menstrual bleeding, and improve mood, as a “natural” antidepressant in the case of severe PMS, although its use in PMS remains controversial.7,8
This is a simplified picture, since the estrogen group actually includes three major hormones: estradiol, estriol and estrone (Fig.1). Estradiol is the predominant estrogen during the reproductive years in terms of absolute serum levels and estrogenic activity. The predominant circulating estrogens are estriol during pregnancy, and estrone during menopause.
Throughout the reproductive years and into menopause, women can also benefit from menstrual cycle mapping. This may involve collecting samples daily to measure fluctuating hormone levels, and hence pinpoint hormone imbalances more accurately than would be possible with a single or more limited number of sampling points. The advent of saliva testing makes accurate mapping more achievable, since it is so much easier to collect saliva samples daily, compared with blood samples.
When a woman arrives at the peri-menopause, and subsequent menopause, the debate rages on as to whether hormone replacement therapy (HRT) is good or bad for her health.5 The answer is a firm: “it depends”, and a practitioner will take into account a patient’s individual history in order to make a recommendation. The main benefits of HRT are that it can help with most menopausal symptoms, such as hot flashes, night sweats, mood swings, vaginal dryness and reduced libido.9 It can also help prevent weakening of the bones (osteoporosis), more common after the menopause.10
Whilst many of these symptoms pass after a few years, they can be unpleasant, and HRT can offer relief for many women. The benefits of HRT are generally believed to outweigh the risks, although one downside is that some types of HRT can increase the risk of breast cancer, so if there is family history of cancer, a woman may simply choose to not take that risk and just ride out the menopausal symptoms instead.9
Hormonal imbalance is also one of the leading causes of female infertility, and with changing hormone levels, a woman’s fertility naturally drops after the age of 35.11 High levels of follicle-stimulating hormone (FSH) can reduce a woman’s chances of getting pregnant, while low levels of luteinizing hormone (LH), which stimulates the ovaries to release an egg and start producing progesterone, can also cause fertility problems. Early menopause and other hormone-related conditions such as PCOS (polycystic ovary syndrome) will also affect fertility.
To confirm a diagnosis of hormone imbalance, the practitioner will first need to establish the levels of the relevant hormones. So how is this done, and what are the market trends as we move towards a more personalized and holistic approach to women’s health?
Measuring female hormone imbalance
Once the symptoms of a potential hormone imbalance have been identified, such as infertility, or hot flashes in menopause, the medical practitioner will first want to measure the level of specific hormones. Typically, blood samples will be taken, and serum-based tests will be performed. However, in the search for less invasive and more cost-effective ways of measuring female hormones, researchers were curious as to whether saliva would be a viable alternative, and saliva-based ELISA hormone testing was born.12
When developing new tests, the result from any test replacing blood sampling must be at least as accurate as the equivalent blood tests, and ELISA-based saliva testing turned out to be a remarkably reliable method for female sex hormone testing, being highly accurate and cost-efficient, as well as painless and convenient for the patient.13 Saliva-based diagnostics are discussed in more detail in this brochure.
Advantages of saliva-based testing
Besides the fact that it is non-invasive and cost-effective, other advantages of saliva-based testing are that it can be carried out anytime, anywhere, and multiple specimens can be collected. Saliva samples are also much easier to store compared with blood samples. In addition, salivary steroids represent the biologically active hormones in the body more accurately than blood or urinary steroids (Fig. 2).

Fig. 2: Steroids in blood and saliva: the case for saliva testing
Given these results, why would a medical practitioner still hesitate in recommending saliva testing over blood sampling? Well, the prevalence of blood testing and the perceived difficulty in replacing and re-validating established assays may be a couple of the hurdles to overcome, but despite this we are seeing a push towards saliva testing, as shown by the evolving market figures.
The market for female hormone testing
Looking at the major application areas outlined above i.e. hormone imbalance associated with menstruation, menopause and infertility, we can glean some indication of how many women might consult and be tested at some point during their lives from an overview of the global prevalence of these issues. Local healthcare infrastructure and cultural issues also need to be considered, since this will affect the number of women who will consult and are able to be treated.
In a systematic review, the pooled prevalence of PMS was found to be 47.8% globally. However, out of an estimated 1.9 billion women of reproductive age, only around 3-5% will have sufficiently severe symptoms (or the economic possibility) to either consult, or self-refer and buy tests online.15-22 Even if only 1% of women consulted and had treatment, this still represents around 20 million individual tests annually.
Infertility affects an estimated 15% of couples globally, amounting to 48.5 million couples. Around 20-30% of these infertility cases are male-related, contributing to 50% of cases overall.23,24 Couples having difficulty conceiving, will systematically need several comprehensive series of hormone tests, whether for diagnosis or subsequent treatment. These may be blood or saliva-based tests.
Tests can also be bought over the counter or online for investigation of menstrual, menopausal, or fertility-related problems. The most common sample types are blood and saliva, with saliva tests becoming increasingly prevalent.13,14 Moreover, in the developed world, our 24/7 online culture means that we are increasingly self-aware, with a trend towards taking personal ownership for our health issues, and internet-based self-diagnosis. This is driving an explosion in the saliva-based point of care (POC) market for checking hormone levels.
Blood to spit: make the switch
Saliva-based testing is a reliable alternative to blood tests, and as such it is enjoying a growth in market acceptance in many application areas, including the female hormone testing market. In our next blog of this series we will explore the ongoing challenges associated with furthering this market acceptance and accelerating the switch from blood to saliva testing.
Subscribe to our blog to stay updated on saliva testing for female hormone imbalance.
References
1. Sufi, S. B., Donaldson, A., Gandy, S. C., Jeffcoate, S. L., Chearskul, S., Goh, H., Hazra, D., Romero, C., & Wang, H. Z. (1985). Multicenter evaluation of assays for estradiol and progesterone in saliva. Clinical chemistry, 31(1), 101–103. PMID: https://pubmed.ncbi.nlm.nih.gov/3965182/ DOI: https://doi.org/10.1093/clinchem/31.1.101
2. Dafopoulos K., Tarlatzis B.C. (2020) Hormonal Treatments in the Infertile Women. In: Petraglia F., Fauser B. (eds) Female Reproductive Dysfunction. Endocrinology. Springer. https://www.springer.com/gb/book/9783030147815
3. Fait T. (2019). Menopause hormone therapy: latest developments and clinical practice. Drugs in context, 8, 212551. PubMed ID: https://pubmed.ncbi.nlm.nih.gov/30636965/DOI: https://doi.org/10.7573/dic.212551
4. Walker, A. (1992) Premenstrual symptoms and ovarian hormones: A review, Journal of Reproductive and Infant Psychology, 10(2), 67-82. Taylor & Francis online: https://www.tandfonline.com/doi/abs/10.1080/02646839208403940 DOI: https://doi.org/10.1080/02646839208403940
5. Lobo, R. Hormone-replacement therapy: current thinking. Nat Rev Endocrinol 13, 220–231 (2017). PubMed ID: https://pubmed.ncbi.nlm.nih.gov/27716751/ DOI: https://doi.org/10.1038/nrendo.2016.164
6. Biggs, W. S., & Demuth, R. H. (2011). Premenstrual syndrome and premenstrual dysphoric disorder. American family physician, 84(8), 918–924. PubMed ID: https://pubmed.ncbi.nlm.nih.gov/22010771/
7. Maybin, J. A., & Critchley, H. O. (2016). Medical management of heavy menstrual bleeding. Women's health (London, England), 12(1), 27–34. PubMed ID: https://pubmed.ncbi.nlm.nih.gov/26695687/ DOI: https://doi.org/10.2217/whe.15.100
8. Dennerstein, L. et al (1985) Progesterone and the premenstrual syndrome: a double blind crossover trial. BMJ, 290, 1617-1621. PubMed ID: https://pubmed.ncbi.nlm.nih.gov/3924191/ DOI: https://doi.org/10.1136/bmj.290.6482.1617
9. Regidor P. A. (2014). Progesterone in Peri- and Postmenopause: A Review. Geburtshilfe und Frauenheilkunde, 74(11), 995–1002. PubMed ID: https://pubmed.ncbi.nlm.nih.gov/25484373/ DOI: https://doi.org/10.1055/s-0034-1383297
10. Tella, S. H., & Gallagher, J. C. (2014). Prevention and treatment of postmenopausal osteoporosis. The Journal of steroid biochemistry and molecular biology, 142, 155–170. PubMed ID: https://pubmed.ncbi.nlm.nih.gov/24176761/ DOI: https://doi.org/10.1016/j.jsbmb.2013.09.008
11. van Noord-Zaadstra, B. M., Looman, C. W., Alsbach, H., Habbema, J. D., te Velde, E. R., & Karbaat, J. (1991). Delaying childbearing: effect of age on fecundity and outcome of pregnancy. BMJ (Clinical research ed.), 302(6789), 1361–1365. PubMed ID: https://pubmed.ncbi.nlm.nih.gov/2059713/ DOI: https://doi.org/10.1136/bmj.302.6789.1361
12. Vining, R. F., McGinley, R., & Rice, B. V. (1983). Saliva estriol measurements: an alternative to the assay of serum unconjugated estriol in assessing feto-placental function. The Journal of clinical endocrinology and metabolism, 56(3), 454–460. PubMed ID: https://pubmed.ncbi.nlm.nih.gov/6822646/ DOI: https://doi.org/10.1210/jcem-56-3-454
13. Belkien, L. D., Bordt, J., Möller, P., Hano, R., & Nieschlag, E. (1985). Estradiol in saliva for monitoring follicular stimulation in an in vitro fertilization program. Fertility and sterility, 44(3), 322–327. PubMed ID: https://pubmed.ncbi.nlm.nih.gov/4029421/ DOI: https://doi.org/10.1016/s0015-0282(16)48855-2
14. Lewis JG. Steroid analysis in saliva: An overview. Clin Biochem Rev. 2006;27(3):139-146. PubMed ID: https://pubmed.ncbi.nlm.nih.gov/17268582/
15. Direkvand-Moghadam, A. et al (2014). Epidemiology of Premenstrual Syndrome (PMS)-A Systematic Review and Meta-Analysis Study. Journal of clinical and diagnostic research : JCDR, 8(2), 106–109. PubMed ID: https://pubmed.ncbi.nlm.nih.gov/24701496/ DOI: https://doi.org/10.7860/JCDR/2014/8024.4021
16. Winer, S. A., Rapkin, A. J. (2006). Premenstrual disorders: prevalence, etiology and impact. Journal of Reproductive Medicine; 51(4 Suppl):339-347. PubMed ID: https://pubmed.ncbi.nlm.nih.gov/16734317/
17. Dennerstein, L., Lehert, P., Bäckström, T. C., & Heinemann, K. (2010). The effect of premenstrual symptoms on activities of daily life. Fertility and sterility, 94(3), 1059–1064. PubMed ID: https://pubmed.ncbi.nlm.nih.gov/19486964/ DOI: https://doi.org/10.1016/j.fertnstert.2009.04.023
18. Dennerstein, L., Lehert, P., & Heinemann, K. (2011). Global study of women's experiences of premenstrual symptoms and their effects on daily life. Menopause international, 17(3), 88–95. PubMed ID: https://pubmed.ncbi.nlm.nih.gov/21903712/ DOI: https://doi.org/10.1258/mi.2011.011027
19. https://www.who.int/reproductivehealth/projects/en/ Accessed 3 June 2020.
20.https://www.un.org/en/development/desa/population/publications/pdf/popfacts/PopFacts_2019-3.pdf Accessed 3 June 2020.
21.https://journals.plos.org/plosmedicine/article?id=10.1371/journal.pmed.1001356 Accessed 3 June 2020.
22. https://data.worldbank.org/indicator/SP.POP.TOTL.FE.ZS Accessed 3 June 2020.
23. https://www.hhs.gov/opa/reproductive-health/fact-sheets/female-infertility/index.html# Accessed 3 June 2020.
24. Agarwal, A., Mulgund, A., Hamada, A., & Chyatte, M. R. (2015). A unique view on male infertility around the globe. Reproductive biology and endocrinology: RB&E, 13, 37. PubMed ID: https://pubmed.ncbi.nlm.nih.gov/25928197/ DOI: https://doi.org/10.1186/s12958-015-0032-1
Keywords:
About the author

Nastya Yeska
Nastya Yeska joined Tecan in 2017 as a product manager responsible for the Complementary medicine product portfolio, including Food intolerance and Saliva diagnostics. Nastya is part of Global Reagent Marketing & Support Department responsible for delivering ELISA solutions. In 2021, she changed her role to Integrated Marketing Manager.













