By Alexandra Sommer
Cardiovascular diseases claim more lives than all forms of cancer combined, accounting for 17.3 million deaths per year. Heart attacks are a primary symptom. The longer it takes to diagnose and treat a heart attack, the greater the damage. But progress in identifying relevant cardiac enzymes and other biomarkers has increased the likelihood of rapid and accurate diagnosis to ensure effective treatment and improve long-term survival. Several cardiac markers have been developed for the rapid diagnosis of patients with chest pain and suspected acute coronary syndrome. One of these markers, cardiac troponin, stands out and the ability to better define troponin levels is expected to help diagnose heart attacks more effectively, and even predict them.
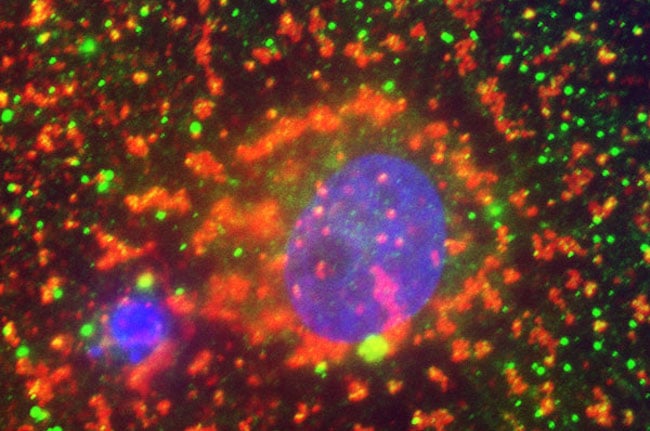
High-sensitivity troponin assays could have far reaching effects on predicting cardiac arrest and reducing healthcare costs. This image shows a cardiomyocyte (cardiac troponin T, green; sarcomeric α-actin, red; DAPI-stained nucleus, blue). Reprinted with permission from AAAS.
The diagnostic value of troponin as a cardiac marker certainly makes it stand out but there is still work to be done to increase the diagnostic accuracy. Meeting the clinical need demands an increase in the sensitivity and precision of troponin assays to shorten the time to diagnosis. It is also crucial to determine the troponin level cut-off point that accurately identifies patients of acute myocardial infarction (AMI) from unaffected individuals.
Troponin: the cardiac marker of choice
A heart attack, or acute myocardial infarction, is the result of insufficient supply of blood to the heart muscle. Cardiac injury that leads to clinical disease occurs when normal cardiac myocyte membranes are disrupted. This causes irreversible damage and releases detectable levels of a number biologically active proteins, or biomarkers, into the extracellular space, including the blood stream.
These biomarkers include troponin, creatine kinase, myoglobin, heart-type fatty acid binding protein, and lactate dehydrogenase. Some of these are used for differential diagnosis of heart conditions, but troponin has become central to the definition of AMI in the consensus guidelines from the European Society of Cardiology and the American College of Cardiology.
Troponin is also used to diagnose post-surgery myocardium trauma, unstable angina, and other diseases related to cardiac muscle injury. An elevated level of troponin may be used as a prognostic indicator to identify patients at high risk for adverse cardiac events.
New players in the diagnostic industry are now developing technologies that are able to detect troponin at very low levels in healthy patients, giving tangible proof that a prognostic test for a heart attack is fully feasible and much cheaper than today’s costly hospital tests, according to Dr. Jeff Bishop of Singulex, a company that has developed a very sensitive test for troponin.
Troponin flags injury earlier
Troponin is a complex of three regulatory proteins (troponin C, troponin I, and troponin T) that are involved in the contraction of skeletal and cardiac muscle. The cardiac sub-forms of troponin I and troponin T can be distinguished from the skeletal forms by immunoassays. Elevated levels can be detected in up to 80% of patients already within 2–3 hours of arriving at the emergency department, in contrast with 6–9 hours for other markers such as creatine kinase. Troponin levels can reach a peak within 16–30 hours, and levels can be elevated even up to eight days after onset of the symptoms, making it possible to diagnose AMI well after the heart attack.
Finding the cut-off
Correct diagnosis depends on reliable cut-off points to identify those patients who are indeed suffering from AMI. The 99th percentile value has become widely used as the reference cut-off to aid in diagnosing AMI. This is the upper limit of what can be expected in a normal, healthy, adult population and it varies from group to group, depending on age and gender.
Guidance from the American Association of Clinical Chemistry on cardiac troponin testing (see Further Reading below) indicates that key components for implementing a cardiac troponin assay include:
- Determining the 99th percentile Upper Reference Limit in a healthy population.
- Ensuring an analytical imprecision of ≤10% coefficient of variation.
- Securing measurement of troponin levels above the limit of detection in ≥50% of healthy subjects.
Since assays are not standardized, this has to be done for each assay. Troponin levels depend on age, especially older than 60 years, and gender, with men having higher levels than women. The AACC recommends determining the 99th percentile values for a minimum of 300 male and 300 female subjects.
Ultra-sensitive immunoassays can change the game
Find out more by watching the presentation by Dr. Jeff Bishop, VP of R&D Singulex: The impact of ultrasensitivity immunoassays: a change in diagnostic modalities.
WATCH THE PRESENTATION
Tecan helps gather samples
When the AACC offered an estimated 11 000 conference delegates the chance to provide blood samples to establish the 99th percentile Upper Reference Limit for cardiac troponin, Tecan was there, providing the robust automation needed for rapid sample handling. Watch the video to find out more.
FURTHER READING
ESC: Acute Myocardial Infarction in patients presenting with ST-segment elevation (Management of) - Clinical Practice Guidelines (Apr 2017)
NICE: Clinical guideline [CG95] - Chest pain of recent onset: assessment and diagnosis - Clinical guideline [CG95] (Nov 2016)
AACC: New guidance on high sensitivity cardiac troponin testing
Cardiac Markers, MedScape
Want to stay updated?
Subscribe to the Tecan blog.
About the author

Alexandra Sommer
Alexandra, Sr. Product Manager Clinical Diagnostics, has a background in oncology and tropical disease research, together with experience in support, marketing and product management in diagnostics. She is enthusiastic about medical advances in assay development and automation technologies that support patient care. Alexandra holds a PhD from the University of Hamburg, with further education in bioinformatics at the Universities of Heidelberg and Mannheim.











