By Nicholas Smith
You are considering an Original Equipment Manufacturing (OEM) partner to support you in bringing your idea to market. The planned in vitro diagnostic device may require components, robotics and modules. You may need integration into an existing platform or the development of a completely new customized system. You may need to react quickly to unexpected circumstances requiring rapid changes in the throughput of your instruments. What else should you take into account when selecting an ideal OEM partner?
Every partnership has two sides and each must work together to reach success. In this case, there is the OEM partner and an OEM customer. Would any OEM partner fit with any OEM customer? There are several success factors that OEM customers and OEM partners need to consider to develop a successful partnership.

Playing to your partners strengths will help to make the most of the relationship.
Keys to a successful OEM partnership
1. Willingness to create a true partnership
Both parties should be willing to adopt a win-win approach when developing an IVD medical device. Win-win means that risks are identified in a transparent manner and are mitigated throughout the program in a spirit of partnership. Clear definition of ownership and responsibilities from every part of the process should be outlined at a very early stage ensuring a well-defined set of deliverables for the optimal IVD medical device. This in turn aligns the expectations of both parties.
A common misconception about OEM development programs is that they finish at product launch. Normally at this point, a good development program moves into life cycle management where manufacturing, supply chain and services are addressed. For this reason, it is important to make sure that both parties are ready and prepared to enter into a lasting relationship, and that the OEM provider is agile enough to react to changes in circumstances such as the need for rapid scale up of the IVD medical device in the event of a surge in demand.
2. Knowledge of the market and good delivery record
As a strong foundation on which to base the definition of requirements, the OEM customer understands the market for in vitro diagnostics and lab automation as well as what the end-user customer needs. In a relationship, the responsibility must be taken to translate those needs into project requirements. Providing regular Voice of Customer (VOC) challenge to the proof of concept, the prototypes, and the final products will confirm that the requirements set are correct and give the confidence that you are developing the right solution for your end-user customers.
The OEM partner should have sufficient knowledge of the in vitro diagnostics market to understand your business and turn your end-user’s lab automation requirements into the appropriate specifications. When selecting an OEM partner, consider whether this potential partner has developed and manufactured similar IVD medical devices or lab automation systems for your target market in the past. Do they have technology that meets the needs of your end-user customers?
3. Full complement of R&D capabilities
A development process is complex and can be a risky journey that requires a variety of complementary technical and supporting skills. Mechanical engineering and software development have to be closely linked with skills in regulation and quality assurance.
Two essential capabilities for an in vitro diagnostics project are the system engineering and integration expertise. Before embarking on the project, ask whether you have a clear understanding of where the gaps are in your in-house capabilities, and then look for an OEM partner that can provide the missing skills and expertise. Additionally, within the partnership, you and your OEM partner can leverage each other’s core competencies, e.g. lab automation (partner) and assay (customer). Co-development, co-feasibility and check points in the program schedule will help de-risk simultaneous assay and system development.
In addition to complementary capabilities, both parties should ensure that there is a well understood assignment of key partner resources. An inefficient use of resources, inadequate selection of skills, or lack of diligence in selecting resources are all important factors which can increase timeline and cost risk.
4. Flexible solutions based on a modular portfolio
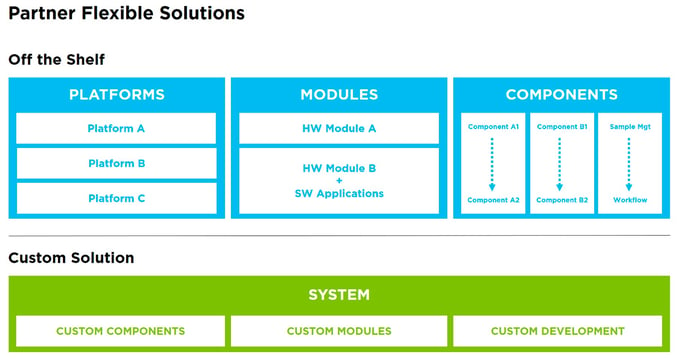
Product technology gaps may result in slower technology readiness or an increased technology learning curve. When selecting an OEM partner, consider what off-the-shelf products they offer that may broaden your own product pipeline. For instance, development of new components, modules, robotics or platforms as initial steps in the development of a full complex system often brings additional risks that can be difficult to mitigate. Integrating already developed and tested elements reduces risk and can lead to faster technology readiness. This applies not only to hardware, but to software as well. Modular software architectures enable faster developments with pretested modular building blocks that can help bring instruments to the market sooner.
5. Flexibility to adopt and adapt to your partner’s process
Maintaining compliant processes and recording all documentation into an internal Design History File (DHF) are key elements of good development practices for the selected OEM partner. Both the OEM partner and OEM customer must practice active joint review of deliverables throughout the OEM development.
Using a mutually agreed risk-based development approach and joint continuous monitoring from the concept stage through feasibility, design, verification, validation and product launch ensures that any deviation from the requirements, the time line, or the budget are readily identified and addressed in a timely fashion.
In addition to the process and know-how, consider the culture of the OEM partner company, specifically how it interacts and collaborates. These factors directly impact how effective the relationship will be. Make sure you are speaking with the same definitions to ensure good communication and share the definition of a successful project. It is also important to think about the launch in the early stages with your OEM partner, so that you can anticipate needs and prepare in advance. For example, you may want to launch in a region where you will need technical support in additional languages. Be sure that your partner can support this.
Is it time to assess what you are looking for in a potential OEM partner? You can use our free checklist to help make sure you cover all the bases.
About the author

Nicholas Smith
Nicholas Smith is Head of Global Marketing and Portfolio Management for Tecan’s Partnering Business. A key function of his team is to work closely with customers to develop new product concepts and proposals based on a thorough analysis of their specific requirements. Nicholas’ joined Tecan in 2012 from Roche Diagnostics where he worked for over 20 years in a variety of roles within marketing and business development.











