By Christian Oberdanner
With “fake news” topping the headlines these days, we’re painfully aware that hearing just part of the whole story can lead to seriously wrong ideas that can have embarrassing or even disastrous consequences. The same is true when analyzing cell populations. Every individual cell has its own story to tell, so population averages and random samples are often misleading. When running assays on a cell imaging system, microplate reader or flow cytometer, can you be sure you are getting the whole truth? If not, it may be time to consider whole-well imaging.

Can you trust your cytometry results? Whole well imaging capability on your microplate reader can help you distinguish fact from fiction.
Every cell counts in cytometry
To get a complete understanding of what’s happing in a cell culture, it makes sense to collect information from all the cells in the sample, without disrupting their behavior and complex interactions. An average intensity measurement made with a microplate reader will not tell you the whole story. Microscope images that capture only a portion of the well will not tell you the whole story. Flow cytometry of individual cells that have been stripped from the culture dish and separated from their neighbors will not tell you the whole story.
Not only can incomplete information be perilously misleading, you may also fail to notice serendipitous occurrences – like a rare interaction between cells that defies current theory, or an emerging clonal population that is expressing unusually high levels of a critical byproduct. Goodbye serendipity, Hello “fake news”!
Do you know what you’re missing?
To get the most from assays performed on adherent cell cultures, you'll want to know what is happening to each and every cell, and where it’s happening—in every well of the plate, and in context with all the other cells in the culture. Otherwise, you are not getting the most out of the sample statistically. Even more importantly, you are likely to miss valuable information about:
- Cell position and distribution – Cells are dynamic entities; they move around, get vital cues from their neighbors, hang together in colonies, and orient themselves toward or away from various stimuli. They rely on spatial information to know when to stop dividing and to function correctly in tissue maintenance, development and regeneration. Image-based information about the location and orientation of individual cells can therefore give you valuable clues about what is happening within the culture as a whole, and can be enabling for many adherent cell assays such as cell migration and wound healing. Because cell position and distribution can vary significantly with time and across the culture surface, the ability to assess the entire cell population at once may be essential to ensure reproducible and reliable results.
- Cell confluence — Accurate assessment of cell confluence is often critical for pinpointing the optimal time window for actions such as initiation of imaging; addition of test compounds, reagents or fluorescent probes; and collection of end-point measurements. Uneven cell distribution or viability across the well can easily skew confluence assessments that are based on population sampling.
- Sub-population responses — Cultured cell populations are typically heterogeneous with respect to many factors including cell cycle phase, gene expression, viability, uptake of fluorescent probes and sensitivity to drugs or stimuli. As a result, population-averaged measures or sampling of only a sub-set of the total population may give misleading or ambiguous results. For example, does a low well-intensity reading collected with a plate reader reflect a high response from a small percentage of cells in the well, or a low-level response from the majority of cells? Are the cells imaged at the center of the well behaving the same as those on the periphery? Without whole-well information, such questions are difficult if not impossible to answer with confidence.
- Rare events and phenotypes — Capturing rare events and cell phenotypes is often essential for applications as diverse as clonal selection, elucidation of molecular mechanisms in disease, and pathogen detection. It is easy to miss rare, transient or localized events if your detection system averages the responses or does not interrogate enough cells in a single pass. For the most statistically robust results, it makes sense to analyze every cell in the sample. This includes taking a look at cell morphology, which can tell you a lot about cell health, viability, function and developmental state.
- Assay quality — A colleague of mine in the pharmaceutical industry once wasted 6 months thinking he had discovered a powerful inhibitor when the truth was that the decrease in assay activity was an artifact due to the overly forceful addition of compound into the well. Why didn’t he notice this sooner? Because he was imaging at the center of the well when the damage was being initiated at the edge, where the liquid dispense head was positioned. This is just one of many examples where looking at the entire well can provide important clues about assay quality, such as edge effects, mixing problems, and more.
The bottom line is that every cell matters, because every cell is telling an important part of the story. If you capture just a few images from an entire well or rely on a population-averaged measurement, you are settling for a smaller effective sample size (read: less robust statistics) and literally throwing away valuable data!
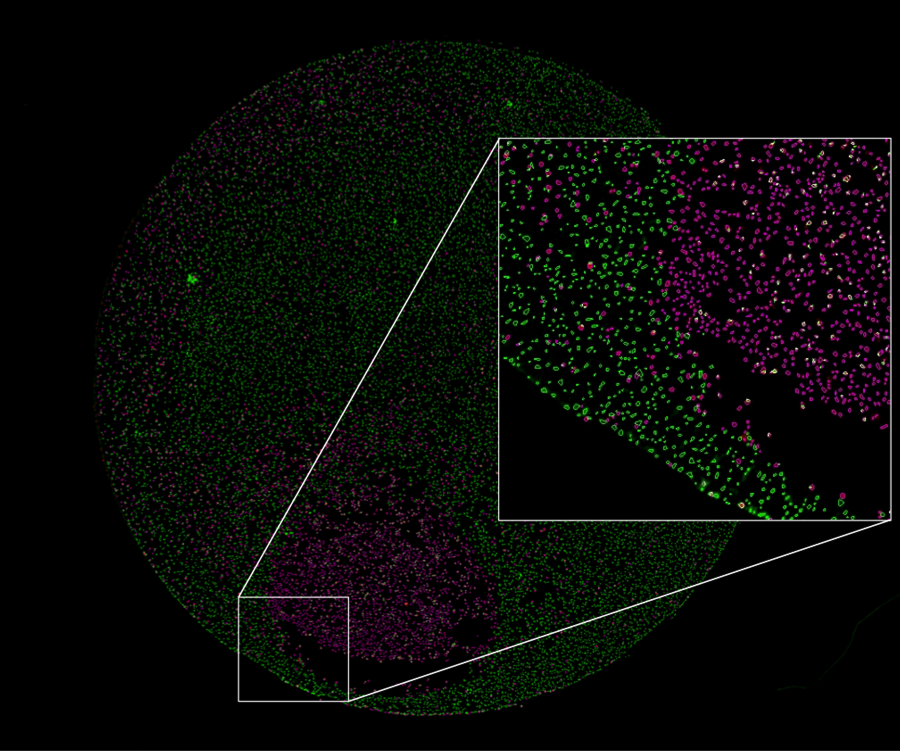
Figure 1. Whole-well fluorescence image from a cell viability assay reveals a discrete patch of dead cells at the well periphery, where a solution containing DMSO was dispensed into the well. Analysis overlays indicate dead cells (magenta overlay) and viable cells (green overlay) detected after staining with propidium iodide and calcein-AM, respectively. Inset shows a close-up of the area demarcated by the white box at the well edge.
Get the whole story with whole-well imaging
The answer? One of the best and least destructive tools for capturing all of the individual cell data from hundreds or even thousands of cells at a time is whole-well imaging (Figure 1). Reliable whole-well imaging, where the entire well area is captured in a single shot, has entered the mainstream only relatively recently. Even more recent is the emergence of whole-well imaging capability for multimode plate readers. Progress has been slow largely because of the technical challenges inherent in achieving sufficient resolution and definition at the well edges, without compromising too much on acquisition speed.
Whole-well imaging increases the power of multimode microplate readers
Fortunately, the technical obstacles are being overcome, and there are now more types of systems on the market that offer whole-well imaging. Multimode microplate readers with whole-well imaging capability are particularly powerful because they give you the freedom to use a broad range of probes and detection modalities. Of course, not all cell imaging platforms perform the task as well or as efficiently as you need them to, particularly if your work involves kinetic analysis of living cells (more on that subject in future blogs). It therefore pays to research what is available on the market and know which factors are critical to enabling high quality whole-well imaging for both fixed and live-cell assays.
If you suspect your cell-based assays are telling you only half the story, check out Tecan's new Spark® Cyto, the first live cell imaging microplate reader to offer real-time cytometry.
Find out more
Learn more about the Spark Cyto live cell imaging microplate reader with real-time cytometry.

About the author

Christian Oberdanner
Dr. Christian Oberdanner is Product Manager at Tecan Austria. He studied molecular biology at the University of Salzburg with a strong focus on cell- and tumor biology during his PhD study. Christian started to work for Tecan Austria as external scientific consultant in 2005 and permanently joined the company in 2006. Since then he held the role of an Application Specialist in the research and development department as well as in the sales and marketing department and since 2015 of a Product Manager. Christian’s priority within Tecan is the multimode microplate reader portfolio.










