By Manuel Bauer
Advances in the treatment of disease, such as the many different types of cancer and cardiac diseases, mean that organ and bone marrow transplantation is on the rise.1 This rise has in turn generated an increased need for accurate immunosuppressant drug (ISD) monitoring. This 3-part blog series will walk you through the challenges of adapting gold standard mass spec methods such as LC-MS/MS for ISD monitoring, and explore ways to avoid the associated pitfalls.

Use LC-MS/MS for accurate immunosuppressant drug monitoring after organ transplantation.
The rise of mass spec methods for drug monitoring
Historically, immunoassays have been the technology of choice for drug monitoring. However, there are serious concerns with immunoassays, including cross-reactivity with metabolites, which can lead to inaccurate drug dosing, as well as sub-optimal quantification performance.2 Increasingly, analysts are looking to replace immunoassays with mass spec (MS) methods such as LC-MS/MS, which offers increased specificity, sensitivity and accuracy over immunoassays. LC-MS/MS is fast, flexible, adaptable, and allows the simultaneous quantification of multiple analytes.3 Nevertheless, successful implementation of LC-MS/MS for ISD monitoring is not without its challenges.
Immunosuppressant drug monitoring: the challenge
The increased number of organ and bone marrow transplants means more people need to be monitored for immunosuppressant drugs (Fig. 1). These drugs reduce the risk for the patient to develop a graft-versus-host disease.
In 2015, more than 75,000 bone marrow transplants were performed globally and the count will have increased by approximately 25% by the end of 2020.4 The increase in bone marrow transplantation is driven by factors such as growing prevalence of blood cancers across the globe, the expansion of bone marrow transplant registries, growing investment in logistic services, and improvement in survival rate after treatment, with a similar story for organ transplantation. 4, 5
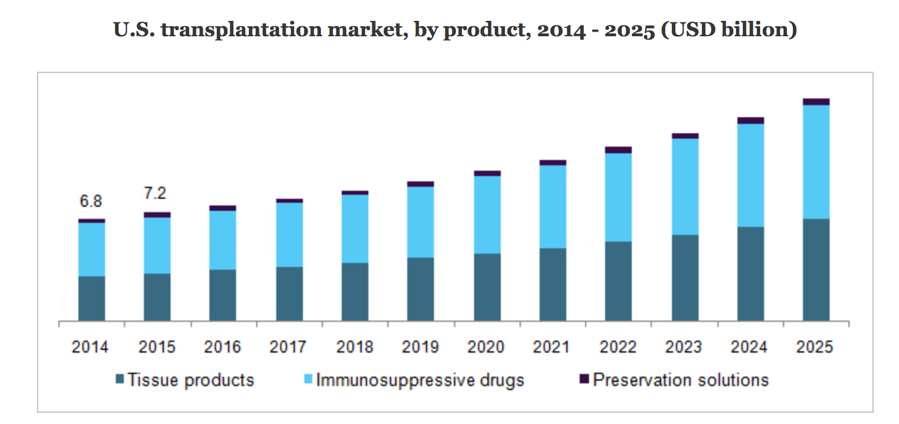
Figure 1. The global increase in organ transplant activities is driving demand for more accurate, rapid and cost-efficient ways to monitor immunosuppressive drugs.
Despite the intrinsic risks of ISDs, their widespread use facilitates the transplantation of tens of thousands of allografts per year, and consequently they have massive potential to decrease patient morbidity and mortality.
Regular and accurate ISD monitoring in these transplant patients is essential to reduce the risks associated with their use, which include:
- Narrow therapeutic range of drug efficacy, with a significant chance of under or over-dosing the patient7
- Variable pharmacokinetics, meaning that a suboptimal amount of the ISD may be available in the patient’s system at any given time
- Potential toxicities if dosage is too high: to other organs (e.g. kidney); via acceleration of diseases like atherosclerosis, diabetes, hypertension; susceptibility to opportunistic infections and malignancies
- Safety and regulatory compliance: strict guidelines must be adhered to for each individual patient
Immunosuppressant drugs are also used to treat autoimmune diseases, in which the immune system attacks the body’s own tissue. Immunosuppressant drugs weaken the immune system, thus suppressing the impact of these harmful autoimmune responses on the body. Autoimmune diseases treated with immunosuppressant drugs include but are not limited to: psoriasis, lupus, rheumatoid arthritis, Crohn’s disease, multiple sclerosis and alopecia areata.8
In our financially-stretched health systems, there is also a world-wide economic drive towards minimizing the cost of monitoring, which competes noisily with the technological drive to individualize and simplify treatment as far as possible, and with the ultimate goal of maintaining the patient’s quality of life throughout and beyond their treatment.
Immunoassays work, so why do we need mass spec?
Although immunoassays (IAs) have been used routinely in ISD monitoring since the ‘80s, their accuracy can be hindered by the metabolites of the ISDs themselves, by antibodies the patient produces against the ISDs, and by heterophilic antibodies, which are endogenous antibodies that bind assay antibodies.9 The consequences of such interference can be devastating.
In the worst case, this cross-reactivity could lead to incorrect dosage of the ISD concerned, and ultimately to organ or bone marrow rejection and patient death. Thus, although IA-based monitoring remains a clinical workhorse, alternative MS-based methods are being developed, in particular LC-MS/MS: liquid chromatography coupled to tandem mass spectrometry. 10
However, LC-MS/MS has had a lot of limitations to overcome, including specific matrix interferences, high purchase and maintenance costs, the need for trained staff, complex method validation, limited robustness, and minimal automation. Also, although LC-MS/MS has superior specificity, allowing it to be less susceptible to interference, its lack of inherent standardization and high throughput modalities have been cause for concern, until fairly recently.
Recent developments include patient-friendly sample submission (e.g. dried blood spots), higher sample throughput and standardization via automation, and the ability to quantify and multiplex ISD-monitoring assays.
These improvements mean clinicians can now use LC-MS/MS to:
- Accurately and efficiently measure blood levels of the ISD they are monitoring
- More easily treat and monitor within the advised therapeutic window for a given patient, both in terms of dosage and timing
- Keep an eye on the effect the ISD may be having on levels of vitamins, hormones and other essential factors
If mass spec is the new gold standard for immunosuppressant drug monitoring, how can we make it less tedious?
Given the challenges outlined above, it is clear that the holy grail for LC-MS/MS success is the elimination of all the variables that are intrinsic to such a complex methodology, beginning with the automation of the sample prep process itself.
To realize their full potential for immunosuppressant drug monitoring, LC-MS/MS methods need to be standardized internationally, and more flexible automation solutions are needed, particularly to address bottlenecks created at the pre-analytical stage of sample preparation.
We will investigate this in our next article in the series. To have the next installments delivered straight to your in-box, subscribe to The Blog now.
References
1. http://www.transplant-observatory.org/ Accessed 19 March 2019.
2. Dasgupta, A. (2016) Personalized Immunosuppression in Transplantation: Role of Biomarker Monitoring and Therapeutic Drug Monitoring. Chapter 2 - Limitations of immunoassays used for therapeutic drug monitoring of immunosuppressants, 29-56.
3. https://www.researchgate.net/publication/47810612_LC-MSMS_for_immunosuppressant_therapeutic_drug_monitoring Accessed 19 March 2019.
4. https://www.persistencemarketresearch.com/market-research/bone-marrow-transplantation-market.asp Accessed 19 March 2019.
5. http://www.transplant-observatory.org/download/2016-activity-data-report/ Accessed 19 March 2019.
6. Hartono, C., Muthukumar, T., & Suthanthiran, M. (2013). Immunosuppressive drug therapy. Cold Spring Harbor perspectives in medicine, 3(9), a015487. doi:10.1101/cshperspect.a015487.
7. Jacob, S., & Nair, A. (2017) A review on therapeutic drug monitoring of the mTOR class of immunosuppressants: everolimus and sirolimus. Drugs & Therapy Perspectives 33(6), 290.
8. https://www.healthline.com/health/immunosuppressant-drugs#qa Accessed 16 March 2019.
9. Steimer.W (1999) Performance and Specificity of Monoclonal Immunoassays for Cyclosporine Monitoring: How Specific Is Specific? Clinical Chemistry, 45 (3), 371-381.
.Aucella, F. Lauriola, V. et al (2013) Liquid chromatography-tandem mass spectrometry method as the golden standard for therapeutic drug monitoring in renal transplant. J Pharm Biomed Anal. 86:123-6. doi:10.1016/j.jpba.2013.08.001. Epub 2013 Aug 11.
10. Feiffer Charão, M. & Linden, R. (2016), Dried blood spots analysis with mass spectrometry: Potentials and pitfalls in therapeutic drug monitoring, Clinical Biochemistry, 49, 1035-1046.
About the author

Dr Manuel Bauer
Dr. Manuel Bauer joined Tecan Switzerland in 2013 and is responsible as Senior Market & Product Manager for Tecan's automated MS sample preparation offering and the Tecan D300e digital dispenser. He studied Biology at the University of Würzburg and Free University of Berlin. During his PhD at the ETH Zürich he focused on systems biology, and has also applied various MS-based proteomics techniques during his post doc at the Biozentrum Basel.











